LifeGene BioMarks
RIPE FOR DISRUPTION
A New Method For Cervical Cancer Screening
At present, women over 30 years old are recommended to schedule cervical cancer screening every three years. In most countries, the gold standard for cervical cancer detection includes Pap smear and HPV tests. Women with an abnormal Pap test and and HPV positive test are referred for a colposcopy-driven biopsy. While a majority of these tests come back negative, biopsies are regularly administered. The current process is often emotionally taxing for women and creates unnecessary health costs.
LifeGene’s precision methylation kits will revolutionize cervical cancer screening. We are developing a non-invasive, streamlined solution for cervical cancer screening and prevention that uses a panel of precision epigenetic markers to identify aggressive cervical disease before a colposcopy-driven biopsy is prescribed. The result? Better prevention, at a lower cost.

LEARN MORE
How It Works
LIFEGENE SOLUTIONS
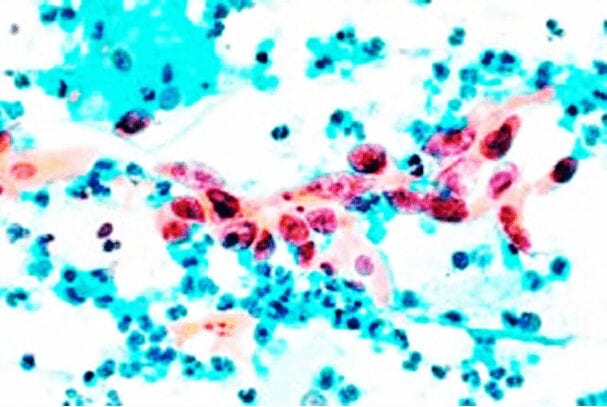
CervicalMethDX Test
Our CervicalMethDX Test is a precision methylation solution that triages HPV positive patients into colposcopy, biopsy or excision procedures.
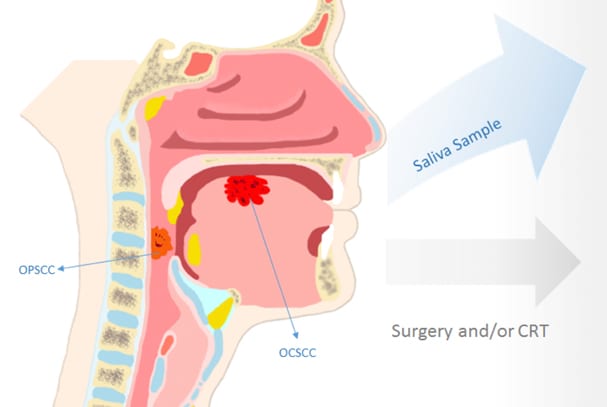
OralMethDX Test
Our OralMethDX Test is a precision methylation solution that screens patients for head and neck cancer.
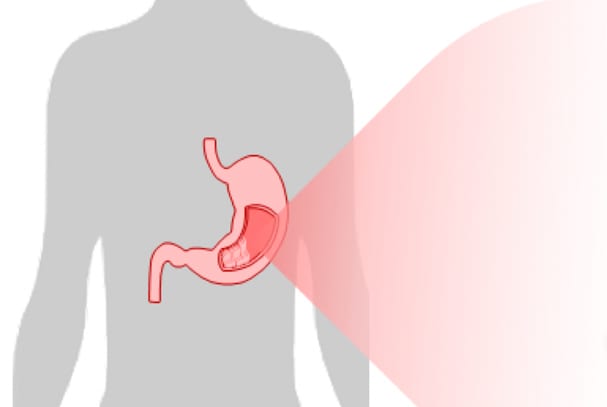
GastricMethDx Test
Our GastricMethDx Test is a precision methylation solution that triages endoscopy-driven biopsies in high-risk gastric cancer clinics.
"We are humanizing cervical cancer screening, globally."
Albert G Thomas MD | Chief Clinical Officer
Better Quality of Life For Everyone, Everywhere.
Connect
Maryland
Garden Level G25P
Baltimore, MD 21205
Puerto Rico
Bo Candelaria
Toa Baja, Puerto Rico 00949